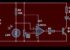
An infrared detector is a detector that reacts to infrared (IR) radiation. The two main types of detectors are thermal and photonic (photodetectors).
The thermal effects of incident IR radiation can be followed by many temperature-dependent phenomena. Bolometers and microbolometers are based on resistance variations. Thermocouples and thermoelements use the thermoelectric effect. Golay cells follow thermal expansion. Pyroelectric detectors are the most common in IR spectrometers.
The response time and sensitivity of photon detectors may be much higher, but they usually have to be cooled to reduce thermal noise. The materials in them are semiconductor with narrow band intervals. The IR photon accident can cause electronic excitation. In photoconductive detectors, the resistivity of the detector element is monitored. Photovoltaic detectors contain a p-n junction on which the photoelectric current is displayed on the illumination.
Infrared or simply Infrared or IR is an electromagnetic radiation (EMR) with wavelengths longer than visible light, and is therefore invisible, although sometimes referred to as generic infrared light. It extends from the red nominal radius of the visible spectrum at 700 nanometers (frequency 430 THz) to 1 millimeter (300 GHz) (although the special pulse lasers allow people to detect IR radiation up to 1050 nm.) The environment is infrared Like all EMR the IRs carry radiant energy and behave both as a wave and as a quantum particle, the photon.
Infrared was discovered in 1800 by astronomer Sir William Herschel, who discovered a type of invisible radiation in the lower energy spectrum compared to red light by its effect on a thermometer. More than half of the total solar energy has ended on Earth in the form of infrared. The balance between infrared radiation absorbed and emitted has a critical effect on Earth’s climate.
Infrared radiation is emitted or absorbed by molecules when it changes its rotating vibrational movements. It excites the vibrational modes in a molecule by changing the dipole moment, making it a useful frequency range for studying these energetic states for the right symmetry molecules. Infrared spectroscopy examines the absorption and transmission of photons in infrared.
Infrared radiation is used in industrial, scientific and medical applications. Night vision devices using near-infrared illumination allow observation of humans or animals without the observer being detected. Infrared astronomy uses sensor-based telescopes to penetrate dusty areas of space such as molecular clouds, detect objects like planets and see objects that have been shaken in red since the early days of the universe. Infrared cameras are used to detect heat loss in isolated systems to observe changes in the bloodstream of the skin and to detect overheating of electrical devices.
Thermal infrared imaging is widely used for military and civilian purposes. Military applications include target acquisition, surveillance, night vision, adaptation, and tracking. People at normal body temperature radiate mainly at wavelengths around 10 μm (micrometers).
Non-military uses include thermal efficiency analysis, environmental monitoring, industrial facility inspections, remote temperature detection, short distance wireless communication, spectroscopy, and weather forecast.