Both isomers have exactly the same atoms in the same order. This means that the dispersion forces between the van der Waals molecules will be the same in both cases. The difference between the two is that the cis-isomer is a polar molecule, while the trans isomer is nonpolar.
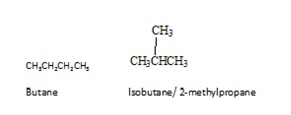
Isomers are different compounds of the same molecular formula. There are different types of isomers. The isomers may be mainly divided into two groups as constitutional isomers and stereoisomers. Constitutional isomers are isomers in which the connectivity of atoms differs in molecules. Butane is the simplest alkane to show constitutional isomerism. Butane has two constitutional isomers, butane itself and isobutene.
In stereoisomers, atoms are connected in the same sequence, unlike constitutional isomers. The stereoisomers differ only in the arrangement of their atoms in space. The stereoisomers may be of two types, enantiomers and diastereomers. Diastereomers are stereoisomers whose molecules are not images mirrored from each other. The cis trans isomers of 1,2-dichloroethane are diastereomers. Enantiomers are stereoisomers whose molecules are non-superposable mirror images from one another. Enantiomers only appear with chiral molecules. A chiral molecule is defined as one that is not identical to its mirror image. Therefore, the chiral molecule and its mirror image are enantiomers of each other. For example, the 2-butanol molecule is chiral, and its image and its mirror are enantiomeric.
As mentioned above, cis trans isomerism or, in other words, E-Z isomerism is a form of stereoisomerism. The Cis transcription system applied to name simple compounds, while the E-Z system is used for more complicated molecules. When a molecule has a restricted rotation at a specific site, the cis and trans isomers may exist.
When there are carbon-carbon double bonds, there is a narrow rotation, so that there may be trans cis isomers. In a molecule such as 1,2-dibromoethene, two bromine groups may be on the same side of the double bond or on the opposite side of the double bond. This does not matter if the link is a single bond, because then the atoms can rotate. However, in this case, two molecules are not the same. Although the Cis and trans isomers have the same molecular formula and molecular weight, their physical properties are different.