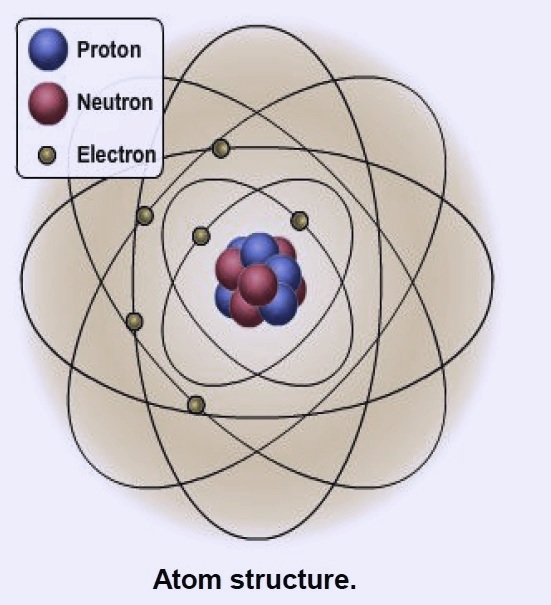
The smallest building blocks of matter are Called atoms. All atoms consist of three Basic components: the proton, the neutron and the Electron See illustration. Different Number and arrangement of protons, Neutrons and electrons give atoms differently Properties that make up one of the more than 100 basic substances known as elements.
Nucleus
The Nucleus is positively charged. It’s the Middle part of an atom and contains protons and neutrons. It makes 99.9% of one Atomic weight. See illustration for Nucleus position.
Proton
The proton is positively charged. The atomic number of an atom is equal to the number of protons in the atom. Atoms have the same number of protons and electrons Atoms electrically neutral.
Neutron
The neutron has no electric charge, which makes it neutral. Lighter elements have over the same number of protons and neutrons. Heavier elements have more neutrons than Protons. Since they have no electrical charge, they no longer make the material positive or negative.
Electrons
Electrons are located at different distances from the nucleus and are arranged in energy levels Called shells or rings. Electrons occupy almost the entire volume of an atom, but Electrons themselves make up only a small fraction of the mass of an atom. Largely the Number of electrons in its outermost ring (commonly known as valence ring) determines the chemical behavior of an atom.
When atoms connect and form Molecules, electrons in the outermost shell, are either transferred from one atom to another or shared between the atoms in a process known as covalent bonding. uniform The movement of electrons in a certain direction is called current flow and can take place used to do work. The electron flow is fundamental for the electrical theory.
Electrical Charge
Electrons are negatively charged particles. Normally, an atom has the same number of Electrons and protons. Each electron carries one negative charge unit each Proton carries a positive charge unit. As a result, the atom is electrically neutral. If An atom gains electrons, it is negatively charged. If it loses electrons, it will positively charged. Electrically charged atoms are called ions.
Valence Ring
The outermost ring determines if a Material will be a conductor or one Insulator. Ladder let electricity flow through them with little or no resistance. Conductors are used to move electricity from place to place. Isolators do not allow Electricity flows easily through them and have a very high resistance. Isolators are used to contain electricity